


Copyright © 2007-2010 BiochemLabSolutions.com
Small molecule structural similarity analysis and clustering
The program for analysis of high-throughput screening data and generally for analysis of data sets containing multiple chemical compounds. This software is most suitable for finding related chemical structures and for establishing structure-activity relationships (SAR) within experimentally generated molecule sets or libraries of small molecules.
The program for analysis of high-throughput screening data and generally for analysis of data sets containing multiple chemical compounds. This software is most suitable for finding related chemical structures and for establishing structure-activity relationships (SAR) within experimentally generated molecule sets or libraries of small molecules.

1. Simple in use.
2. Fast performance.
3. The chemical similarity searches go beyond detection of just a common scaffold. The program can find
non-obviously related compounds. To learn more, read about Tanimoto scoring and Open Babel.
4. Easy DragDrop-based SDF clustering with minimum tuning or setup steps.
2. Fast performance.
3. The chemical similarity searches go beyond detection of just a common scaffold. The program can find
non-obviously related compounds. To learn more, read about Tanimoto scoring and Open Babel.
4. Easy DragDrop-based SDF clustering with minimum tuning or setup steps.
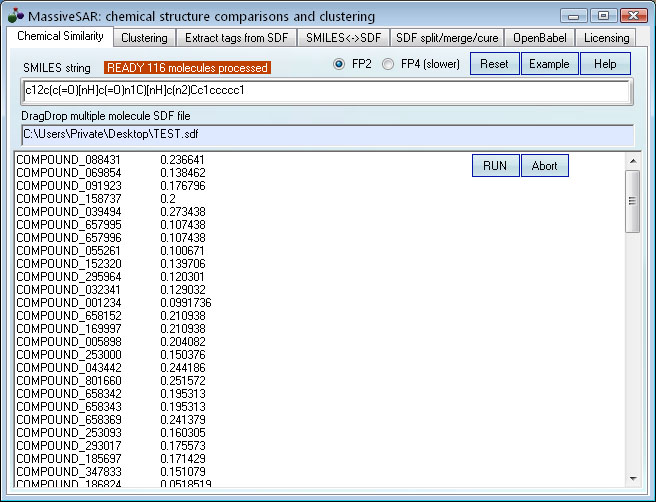
The input: a SMILES string and an SDF database containing many small molecules.
The output: a list of similarity indexes between the SMILES query and each molecule in the SDF file.
The output: a list of similarity indexes between the SMILES query and each molecule in the SDF file.
SDF tag extraction utility
SDF split, merge & repair utility

MassiveSAR
small molecule analysis and clustering functions
- Find molecules in SDF databases by SMILES string
- Cluster any SDF datasets by related chemical scaffolds
- Work with and sort SDF databases by SDF tags (such as hit scores, MW, etc)
- Add new SDF tags to SDF databases
- Extract SDF structures of interest from large SDF databases by using structure names or tags
- Extract SDF tags
- Split/Merge SDF data sets
- One-step SDF <-> SMILES conversion for any dataset sizes
small molecule analysis and clustering functions
- Find molecules in SDF databases by SMILES string
- Cluster any SDF datasets by related chemical scaffolds
- Work with and sort SDF databases by SDF tags (such as hit scores, MW, etc)
- Add new SDF tags to SDF databases
- Extract SDF structures of interest from large SDF databases by using structure names or tags
- Extract SDF tags
- Split/Merge SDF data sets
- One-step SDF <-> SMILES conversion for any dataset sizes











